In 2005 Dr. Anthony Fauci,the director of the (NIAID) National Institute of Allergy and Infectious Diseases, a position he still holds received the encouraging news that Chloroquine is a potent inhibitor of SARS coronavirus infection and spread. The SARS epidemic petered out, ao the clinical trial was never executed.
Now the news about the efficacy of HydroxyChloroquine is coming in hot and heavy. The results are very encouraging. This is from a recent study in Marseilles, France:
We retrospectively report on 1061 SARS-CoV-2 positive tested patients treated for at least three days with the following regimen: HCQ (200 mg three times daily for ten days) + AZ (500 mg on day 1 followed by 250 mg daily for the next four days). Outcomes were death, clinical worsening (transfer to ICU, and >10 day hospitalization) and viral shedding persistence (>10 days).
Results
A total of 1061 patients were included in this analysis (46.4% male, mean age 43.6 years – range 14–95 years). Good clinical outcome and virological cure were obtained in 973 patients within 10 days (91.7%). Prolonged viral carriage was observed in 47 patients (4.4%) and was associated to a higher viral load at diagnosis (p < .001) but viral culture was negative at day 10. All but one, were PCR-cleared at day 15. A poor clinical outcome (PClinO) was observed for 46 patients (4.3%) and 8 died (0.75%) (74–95 years old). All deaths resulted from respiratory failure and not from cardiac toxicity. Five patients are still hospitalized (98.7% of patients cured so far). PClinO was associated with older age (OR 1.11), severity of illness at admission (OR 10.05) and low HCQ serum concentration. PClinO was independently associated with the use of selective beta-blocking agents and angiotensin II receptor blockers (p < .05). A total of 2.3% of patients reported mild adverse events (gastrointestinal or skin symptoms, headache, insomnia and transient blurred vision).
Conclusion
Administration of the HCQ+AZ combination before COVID-19 complications occur is safe and associated with a very low fatality rate in patients.
Since then, prescriptions for the drug cocktail in Marseilles is up 7000%, and is also increasing in the rest of France, especially the Paris region.
But France is by no means the first country to go all out prescribing the drug cocktail. At least twelve countries are doing it, and Turkey and Morocco prescribe it to all with COVID-19 symptoms ( after first checking their heart). Russia joined six weeks later. Here are the results:
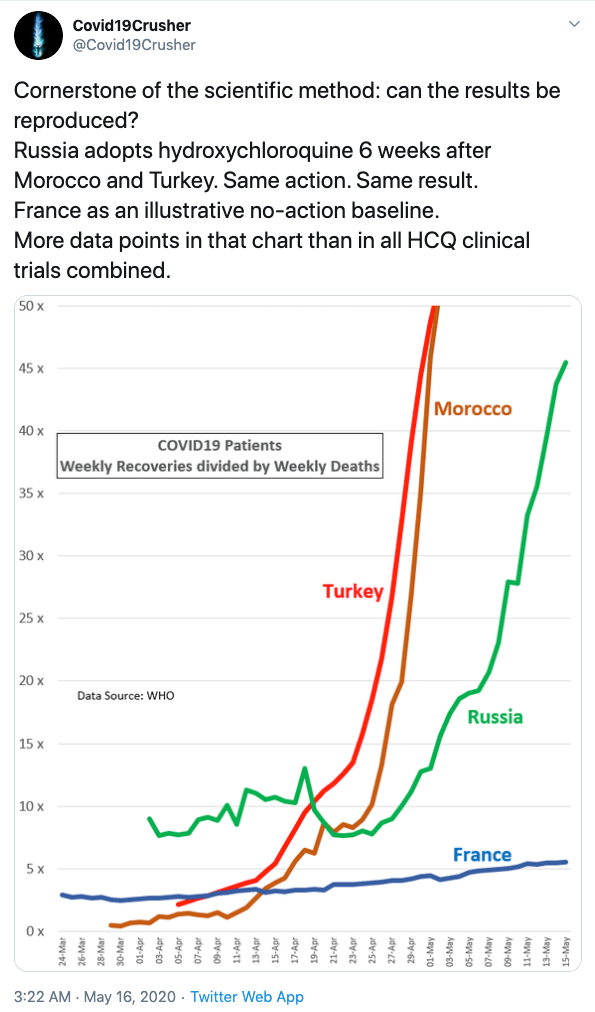
If we look at the results from onset to today, some interesting facts stand out
Turkey: Death rate 50 per million. ratio of recovered cases/death is 37
Morocco: Death rate 5 per million. ratio of recovered cases/death is 19.6
Russia: Death rate 19 per million. ratio of recovered cases/death is 19.6 and rising, but they started six weeks later and have not yet reached their infection maximum.
France: Death rate 433 per million. ratio of recovered cases/death is 2.2, but their new case maximum was 11 weeks ago and the maximum death rate was 9 weeks ago, way before any prescriptions increased.
U.S. does not do an even job of reporting recovered cases, so the results will only be valid for some states. Michigan seems to be one state that records recoveries.
Michigan: Death rate 492 per million. ratio of recovered cases/death is 5.7
The best state is South Dakota, but there Governor Kristi Noem conducted a semi clinical (not double blind) trial, and the results speak for themselves.
South Dakota: Death rate 50 per million. ratio of recovered cases/death is 63.27 !!
My suggestion is this: Give the HCQ+ AZT + Zinc +Vitamin D for 5 days as soon as symptoms occur. Give HZQ + Zinc + Vitamin D for 5 days to all the infected person’s contacts, then their quarantine is over, otherwise it is 14 days. Check for heart rhythm problems, but otherwise go ahead.
The results from Turkey + Russia + Morocco involved more than 170,000 recovered cases, far more than any clinical study, and showed the effect both before and after HCQ became the drug of choice, so the effect is real enough!

