The HCQ test made in Qatar
is stunning, that is all I can utter
For the death rate is low
A great fearmonger blow
Ignore it! The media mutter.
Qatar is an interesting country. It hosts America’s biggest Air base in the Middle East. With a population of only 2,8 million people it has 45,465 people that tested positive for the coronavirus, or 1.58% of the population, the highest in the world reported so far, bur only 26 deaths so far, only 0.009%, and there are nearly 400 recoveries for every death, the highest in the world.
How can that be? Does Qatar have a very unusual population profile? Yes, indeed
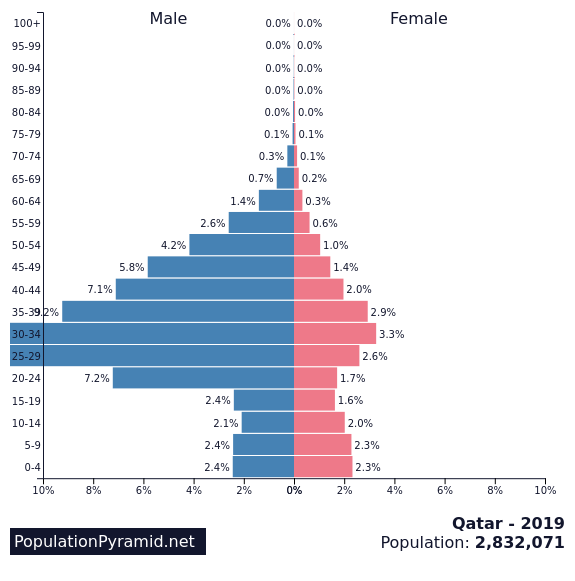
but it is not because of many children, no, over half of the population is working age males, nearly all guest workers. This explains a lot, but even that is not the main cause.
No, they have done a nearly universal medical protocol for all that test positive for the COVID-19 virus. All will get Hydroxychloroquine + Azithromycin , but a random sample will be selected for a small, double blind study. It started April 14 and ended May 14. The results will be published May 30 of so, but we don’t have to wait for the results of that test to verify the effectiveness of this medical treatment if begun as soon as symptoms materialize. To have the highest infection rate and the lowest death rate in the world is sufficient reason for me to advocate HCQ + AC for all eligible testing positive. Maybe if you include Zinc in the protocol the results would be even better, but we cannot wait for all the results to come in. Let us save lives now!
Appendix: Some were not be eligible for this study. The exclusion criteria were:
| Ages Eligible for Study: | 18 Years and older (Adult, Older Adult) |
| Sexes Eligible for Study: | All |
| Accepts Healthy Volunteers: | No |
Inclusion Criteria:
- Patient is in HMC facility for low-acuity, Covid-positive patients being quarantined.
- Positive Covid test on qualitative assay used during routine care (i.e. not as part of Q-PROTECT (that is: victims given the placebo to make it a true random test)
- Age at least 18
Exclusion Criteria:
- Treating physician judges patient not appropriate for study participation for any reason
- Age <18
- Breastfeeding or pregnancy (patient-reported pregnancy status is sufficient)
- Hypersensitivity to chloroquine or HC or AZ
- History of or known QT prolongation
- EKG required before study entry and on each visit during the subject’s first seven days on pro-tocol, during the time period HC is being taken
- Baseline QTc >480 if QRS width normal; QTc >510 if QRS >120
- Known G6PD deficiency, porphyria, or retinopathy (eye exam prior to study entry)
- Known hepatic or renal disease (or abnormality on liver or renal function testing at study day 1)
- Low magnesium or low potassium (by testing on day 1)
- Current (pre-study) therapy with antimalarial or dapsone
- Current (pre-study) therapy with antiviral agents (e.g. oseltamivir)
- Tisdale36 score exceeding 6 as tallied below (based on ACC recommendations)*
- 1 point each: age>67, female sex, or being on loop diuretic
- 2 points each: serum potassium <3.6, QTc>449, acute myocardial infarction
- 3 points each: sepsis, heart failure, QT-prolonging drugs